 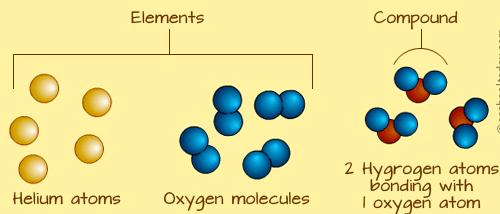
The electrons are shared between the two elements to fill the outer shell of each, making both elements more stable. To completely fill the outer shell of oxygen, which has six electrons in its outer shell but which would be more stable with eight, two electrons (one from each hydrogen atom) are needed: hence the well-known formula H 2O. The electron from the hydrogen splits its time between the incomplete outer shell of the hydrogen atoms and the incomplete outer shell of the oxygen atoms. The hydrogen and oxygen atoms that combine to form water molecules are bound together by covalent bonds. 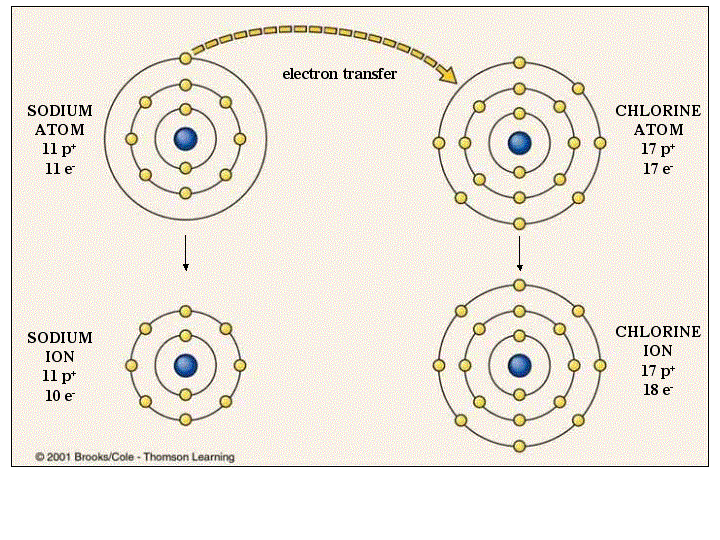
The formation of water molecules provides an example of covalent bonding. This strong triple bond makes it difficult for living systems to break apart this nitrogen in order to use it as constituents of proteins and DNA. Molecular nitrogen consists of two nitrogen atoms triple bonded to each other and, as with all molecules, the sharing of these three pairs of electrons between the two nitrogen atoms allows for the filling of their outer electron shells, making the molecule more stable than the individual nitrogen atoms. The strength of different levels of covalent bonding is one of the main reasons living organisms have a difficult time in acquiring nitrogen for use in constructing their molecules, even though molecular nitrogen, N 2, is the most abundant gas in the atmosphere. The more covalent bonds between two atoms, the stronger their connection. One, two, or three pairs of electrons may be shared, making single, double, and triple bonds, respectively. Covalent bonds are also found in inorganic molecules like H 2O, CO 2, and O 2. Covalent bonds are commonly found in carbon-based organic molecules, such as our DNA and proteins. These bonds are stronger and much more common than ionic bonds in the molecules of living organisms. Describe the characteristics of covalent bonds and differentiate between polar and nonpolar bondsĪnother way the octet rule can be satisfied is by the sharing of electrons between atoms to form covalent bonds. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
